Lucialdehyde C
IUPAC name: E,6R)-6-[(3S,5R,10S,13R,14R,17R)-3-hydroxy-4,4,10,13,14-pentamethyl-7-oxo-1,2,3,5,6,11,12,15,16,17-decahydrocyclopenta[a]phenanthren-17-yl]-2-methylhept-2-enal
CAS Number: 252351-96-5
Class: Terpenoid
Mushrooms contain this compound
1 mushrooms
| MushID |
Thai name |
Common name |
Scientific name |
Family |
| 1 |
เห็ดหลินจือ |
Lingzhi |
Ganoderma lucidum ( Curtis ) P. Karst. |
Ganodermataceae |
2D Structure
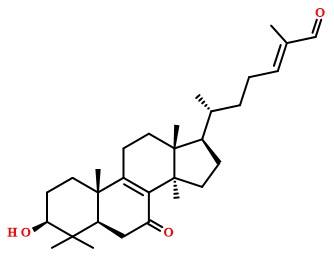
Compound Properties
SMILES
O=C/C(=C/CC[C@H]([C@H]1CC[C@@]2([C@]1(C)CCC1=C2C(=O)C[C@@H]2[C@]1(C)CC[C@@H](C2(C)C)O)C)C)/C
Physicochemical Properties
| Formula |
C30H46O3 |
| Molecular wieght(g/mol) |
454.68 |
| Num. of Heavy atoms |
33 |
| Num. of Aromatic Heavy atoms |
0 |
| Fraction Csp3 |
0.80 |
| Num. of Rotatable bonds |
5 |
| Num. of H-bond acceptors |
3 |
| Num. of H-bonds donors |
1 |
| Molar Refractivity |
137.44 |
| TPSA (square Angstrom) |
54.37 |
Lipophilicity
| iLOGP |
4.23 |
| XLOGP3 |
6.62 |
| WLOGP |
6.84 |
| MLOGP |
4.82 |
| Silicos-IT LOGP |
7.20 |
| Consensus LOGP |
5.94 |
Water Solubility
| ESOL |
|
| LogS |
-6.50 |
Solubility(mg/ml) |
1.44E-04 |
Solubility(mol/l) |
3.16E-07 |
Class |
Poorly soluble |
| Ali |
|
| LogS |
-7.56 |
Solubility(mg/ml) |
1.24E-05 |
Solubility(mol/l) |
2.74E-08 |
Class |
Poorly soluble |
| Silicos-IT |
|
| LogS |
-6.62 |
Solubility(mg/ml) |
1.09E-04 |
Solubility(mol/l) |
2.40E-07 |
Class |
Poorly soluble |
Pharmacokinetics
| GI absorbtion |
High |
| BBB permeant |
No |
| Pgp substrate |
No |
| CYP1A2 inhibitor |
No |
| CYP2C19 inhibitor |
No |
| CYP2C9 inhibitor |
No |
| CYP2D6 inhibitor |
No |
| CYP3A4 inhibitor |
No |
| Log Kp (skin permeation) (cm/s) |
-4.37 |
Druglikeness
| Lipinski Num. violations |
1 |
| Ghose Num. violations |
3 |
| Veber Num. violations |
0 |
| Egan Num. violations |
1 |
| Muegge Num. violations |
1 |
| Bioavailability Score |
0.55 |
Medicinal Chemistry
| PAINS Num. of alerts |
0 |
| Brenk Num. of alerts |
2 |
| Leadlikeness Num. of violations |
2 |
| Synthetic Accessibility |
5.95 |
Biological Activity
| Biological Activity |
JID |
| Antitumour |
7 |
| Anti-HIV |
7 |
| Hypoglycemic |
7 |
| Antiplatelet aggregation |
7 |
| Neuroprotective |
7 |
| Hepatoprotective |
7 |
| Antiobesity |
7 |
